Sulphuric Acid Manufacturing Process
Sulphuric Acid, also known as Vitriol is composed of elemental sulphur, Hydrogen and Oxygen.
The molecular formula is :
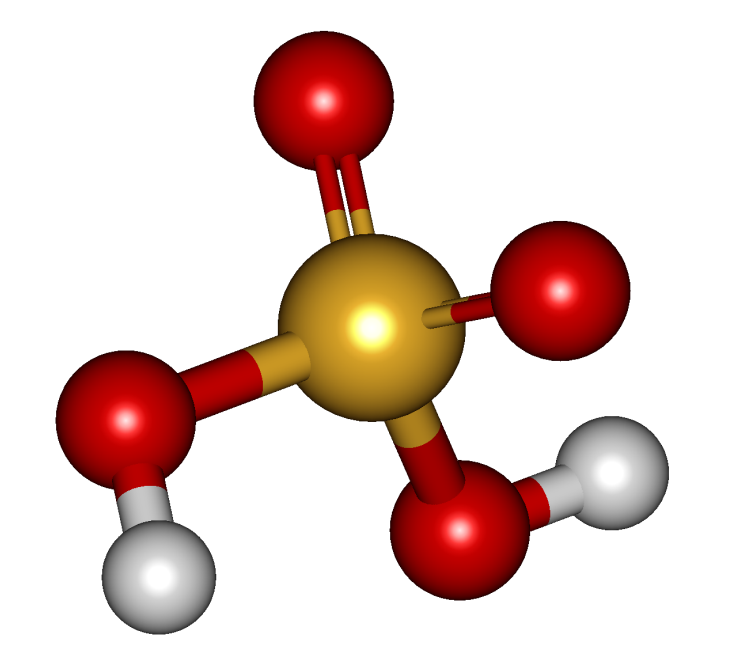
Physical Characteristics : Colourless; Odorless; Soluble in water and is highly exothermic in nature. Considered as Strong Acid.
Chemical Characteristic : Highly corrosive in nature, Hygroscopic, Produces severe chemical and thermal burns when in contact.
Aslo Read This :
Vinyl Chloride from Ethylene
Urea Production and Manufacturing
Cement Manufacturing Process
Industrial Sulphuric Acid Production
In industry sulphuric acid is mainly produced by
- Contact Process :
- Wet Sulphuric Acid Process
- Lead Chamber Process : Produces Dilute Sulphuric Acid (67-78%
)
Chemical Reactions Involved :
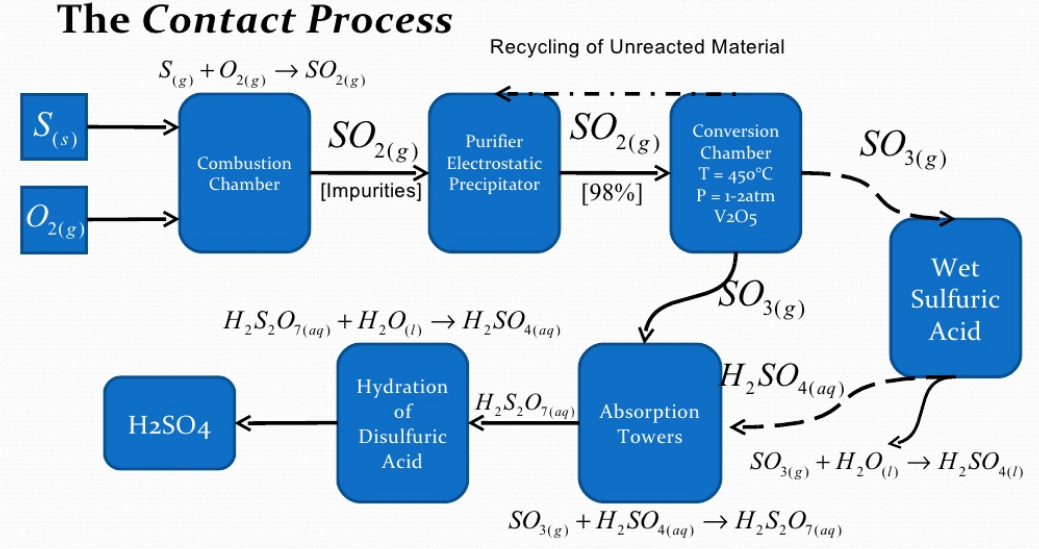
Step 1 : Sulphur is burned to produced Sulphur Oxide
Step 2 : Then Sulphur Oxide is oxidized to sulfur trioxide using oxygen in the presence of a vanadium(V) oxide catalyst . This formation of sulphur trioxide is a reversible and highly exothermic reaction.
The sulfur trioxide is absorbed into 97–98% to form oleum (
), which is also known as fuming sulfuric acid. Thenafter oleum (
) is diluted with water to form concentrated sulfuric acid
.
Sulphuric Acid Production Process Flow Sheet
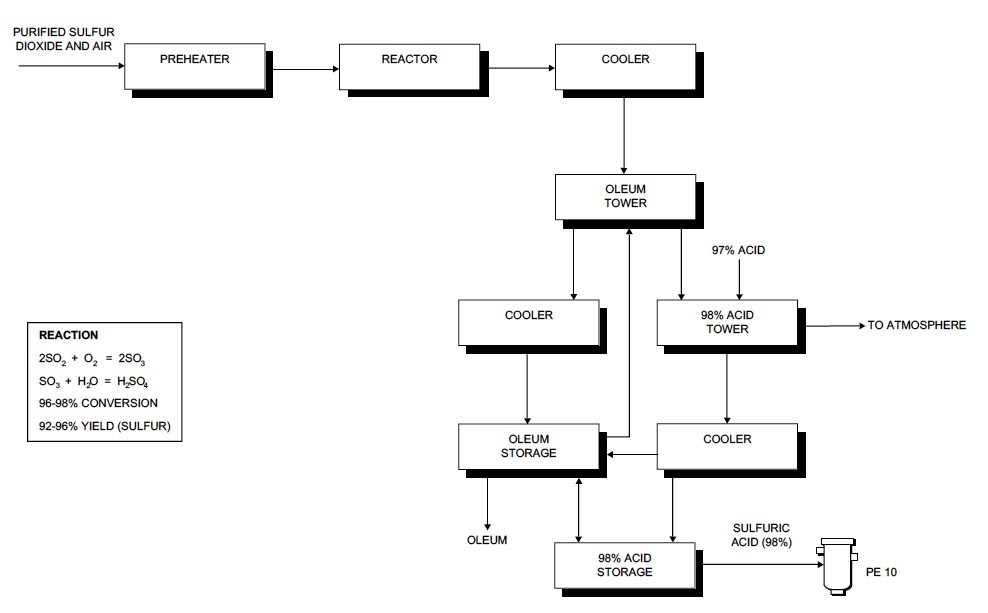
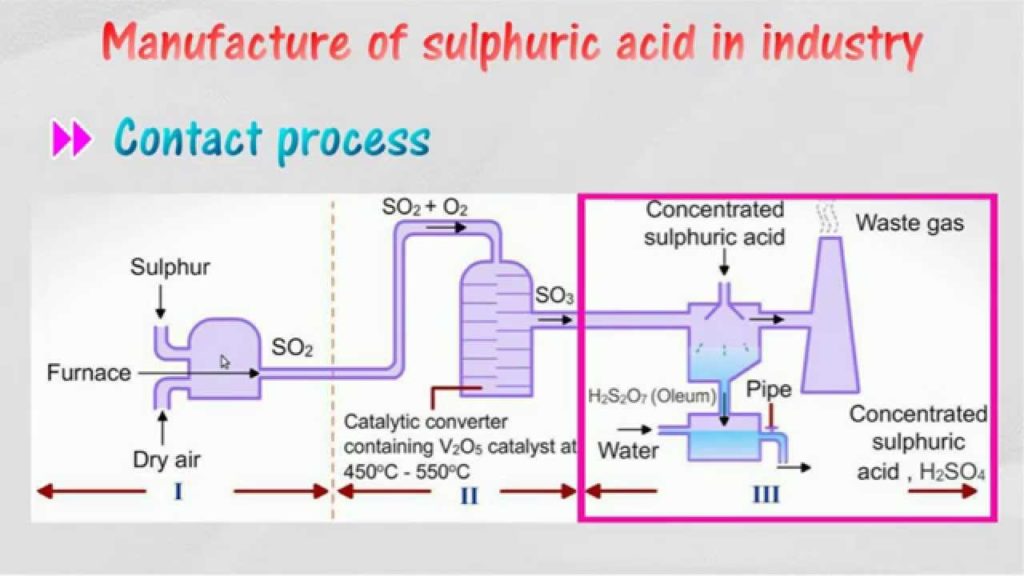
Industrial Sulphuric Acid Plant

Sulphuric Acid Applications
- Paint & Pigments
- Pharma & Bio- Pharmaceuticals Industries
- Fertilizer Manufacturing
- Chemical Synthesis
- Uses in Oil and Waste water processing Industries
- Manufacturing Cleaning Agents
- Batteries
- Plastic Industry
ImageSource : Wikipedia; askIITians; DuPont;
ArticleSource : Wikipedia

















1 Response
[…] Sulphuric Acid Manufacturing Process […]