What Is Triple Point ?
What is Triple Point ?
The Triple is considered as a thermodynamic equillibrium point. At this point all the three states of an element; Solid, Liquid & Gas form considered at an equillibrium. Triple point is different for different system.
Example, Triple point of Acetylene occurs at -80.7° Celsius.
Triple point of any substance is a single point at a peculiar temperature and pressure. It is the point where all the three phases coexists at equillibrium.
For Example : Consider water as a system. The triple point of water is at 273.16 K and the assosiated pressure is 611.2 Pa.
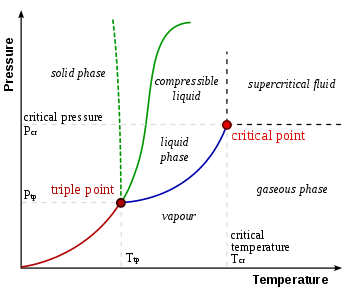
Similarly, For Carbon Dioxide the triple point coexist at 5.2 atm (3952 torr) and 216.6 K (-56.4oC).
Read Also :
Phase Change Nomenclature
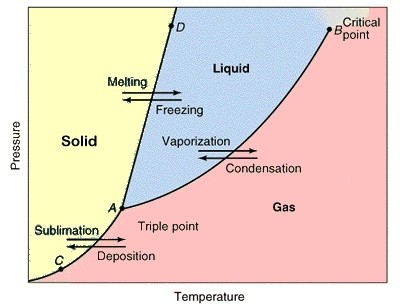
Solid to Liquid : Melting
Liquid to Solid : Freezing
Liquid to Gas : Vaporization
Gas to Liquid : Condensation
Solid to Gas : Sublimation
Gas to Solid : Deposition
Data Source : Chemistry LibreTexts;
ImageSource : Wikipedia; UW-Platteville