Nitric Acid Manufacturing
Nitric Acid () is also known as aqua fortis or Spirit of niter or Hydrogen nitrate.
Physical Characterestics :
- Highly corrosive in nature
- Appearance : Colorless, yellow or red fuming liquid
- Odor : Suffocating in smell
- Solubility : Highly miscible in water
- Molar mass : 63.01 g/mol
Read Also :
Sulphuric Acid Manufacturing Process
Urea Production and Manufacturing
Chemical Characteristics :
- Considered as strong oxidizing agent
- Thermally sensitive : Explosive in nature
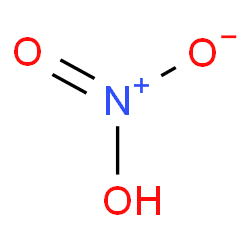
- Chemical Structure
In industry nitric acid is manufactured by Ostwald process. This process of making nitric acid was developed by Wilhelm Ostwald.
Ostwald process is very close to Haber process which is used for the manufacturing of Ammonia.
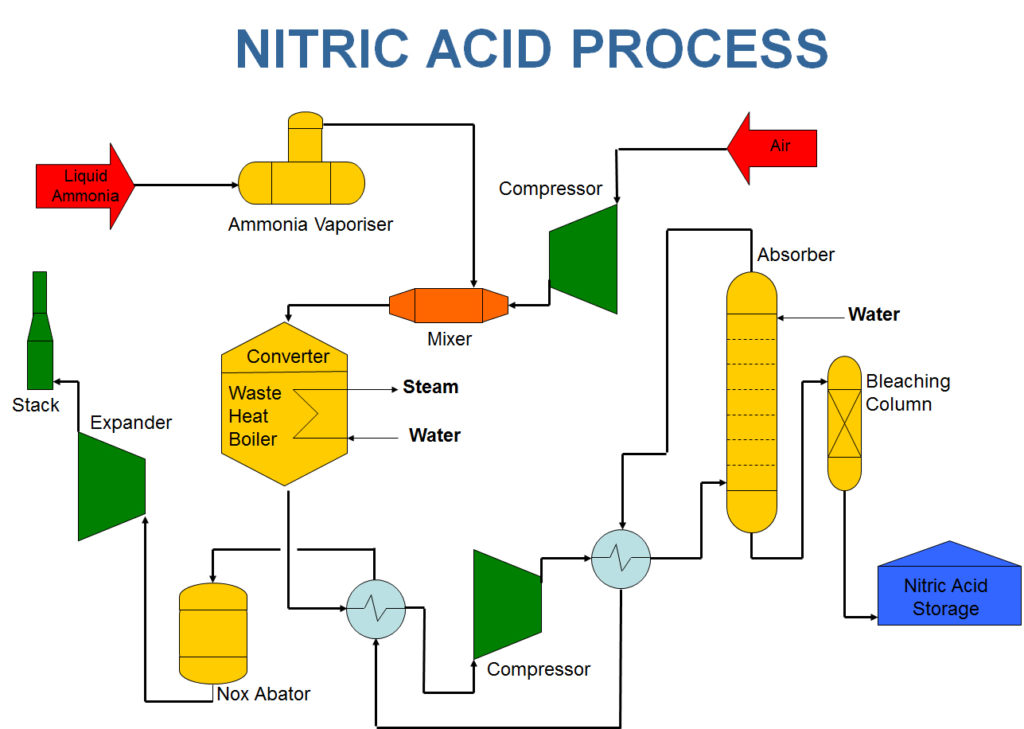
- For the manufacturing of Nitric Acid, Ammonia is used as a prime raw material and platinum + 10% rhodium is used as a catalyst.
Description :
Ammonia is first oxidized to nitric oxide in presence of heat, oxygen and the catalyst. This reaction is highly exothermic in nature.
Operating Parameters :
- Pressure : 4–10 standard atmospheres (410–1,000 kPa; 59–150 psi)
- Temperature : 870–1,073 K (600–800 °C; 1,100–1,500 °F)
In second step this nitric oxide is further oxidized (leading to the formation of nitrogen dioxide ) and afterwards is getting absored in water by using an absorption appratus. This leads to the formation of Nitric acid which is in diluted form.
For getting concentrated Acid this NO is further recycled and distillated.
This is the brief mechanism of manufacturing of nitric acid in industry.
There is a no. of applications of Nitric Acid () such as :
- In the formation of ammonium nitrate
- Frequently used in the production of fertilizers
- In the nitration of glycerol and toulene
- In the formation of Explosives such as nitroglycerine and trinitrotoulene (TNT)
- In the preparation of Nitro-cellulose
- In polymers (eg: Nylon)
ImageCredit : ChemSpider